INTRODUCTION
Seamounts are isolated topographic elevations rising at least 1,000 m from the seafloor, with summits often a few hundred meters below the surface or within the photic zone (Wessel et al., 2010; Yesson et al., 2011; Rogers, 2018). The thousands of seamounts that occur throughout Earth’s ocean display diverse physical characteristics such as slope, height, and summit depth. By interacting with ocean currents, seamounts modify conditions at local and global scales, creating biological hotspots in otherwise low-productivity areas of open ocean (Morato et al., 2010; Wessel et al., 2010; Rogers, 2018). Consequently, seamounts can aggregate phytoplankton, zooplankton, micronekton, and fish, thus attracting predators (Lavelle and Mohn, 2010; Cascão et al., 2017; Denda et al., 2017; Rogers, 2018; Leitner et al., 2020). Marine megafauna, such as sea turtles, marine mammals, seabirds, elasmobranchs, and commercially important fish including tunas, often aggregate at seamounts (Morato et al., 2010, 2016). Despite increasing knowledge of seamount ecosystems, understanding which characteristics attract marine megafauna remains challenging, especially considering that not all seamounts are hotspots of biodiversity or trophic interactions (Leitner et al., 2020). Addressing this gap would effectively protect productive and sensitive seamount sites in the High Seas (Tiller et al., 2019).
Increasing anthropogenic threats to seamount ecosystems make their protection imperative (Rogers, 2018). Bottom trawling and demersal longlining disturb benthic and pelagic communities (Koslow et al., 2000; Althaus et al., 2009; Clark and Rowden, 2009). Additional threats include deep-sea mining, marine pollution, and climate change impacts that may alter seamount ecosystems’ physical and chemical properties (Rogers, 2018; Washburn et al., 2023). These threats compound broader population challenges affecting marine megafauna that utilize seamounts (Veit et al., 1997). Models predict rising extinction rates from threats including bycatch, overfishing, ship collisions, pollution, environmental changes, and habitat degradation (McCauley et al., 2015; Pimiento et al., 2020). As top predators affecting lower trophic levels and nutrient flows (Hunt and McKinnell, 2006; Doughty et al., 2016), these species serve as ecosystem indicators; they provide insights into environmental processes and help to define priority conservation areas that support diverse marine life and contribute to global biodiversity and ecosystem function (Hazen et al., 2019).
Seabirds are particularly valuable bioindicators for marine ecosystems (Frederiksen et al., 2007; Oppel et al., 2018). Their longevity, wide-ranging foraging, and sensitivity to changes in prey abundance and distribution (including vertically migrating mesopelagic prey), coupled with research accessibility at breeding colonies, make them ideal for detecting oceanic changes (Frederiksen et al., 2007; Hazen et al., 2019). Analyzing seabird tracking data relative to seamount characteristics can help reveal which seamounts attract marine megafauna and the mechanisms that drive hotspot patterns (Oppel et al., 2018; Neves et al., 2023). Tracks often define priority conservation areas, such as marine Important Bird and Biodiversity Areas (Arcos et al., 2012), which can lead to marine protected area (MPA) establishment (Davies et al., 2021). This is particularly important for declining oceanic seabird species like albatrosses and petrels (Procellariiformes) that use seamounts as foraging oases and are at high risk of fisheries bycatch in productive yet unprotected areas (Morato et al., 2008b).
Efforts are underway to establish MPA networks, including seamount ecosystems (Clark et al., 2011). Effective seamount conservation requires international cooperation and development of robust monitoring, control, and surveillance (MCS) strategies to cover remote regions (Gubbay, 2003). The new Agreement under the United Nations Convention on the Law of the Sea (UNCLOS) regarding the conservation and sustainable use of marine biological diversity of areas beyond national jurisdiction (the “BBNJ Agreement”) helps to address MCS strategies by providing mechanisms for closing scientific, financial, technical, and policy gaps for States with limited MCS capacity in such areas (Cremers et al., 2020). Growing international attention to deep sea governance is reflected in two International Union for the Conservation of Nature (IUCN) resolutions adopted in October 20241 calling for seamount protection from destructive fishing practices and advocating precautionary management of the mesopelagic zone (Morgan and Bent, 2025). Such conservation frameworks depend on identifying specific features that make seamounts productive, yet gaps remain in understanding which factors drive these patterns.
We review existing knowledge on seamount features that enhance productivity and attract marine predators, focusing on seabirds, as fine-scale spatial data on them are abundant. These characteristics may promote ecological activity and biodiversity, serving as metrics for determining area attractiveness for predators (Figure 1). The factors that drive this attraction can in turn serve as conservation tools. We focus on the Cabo Verde and Azores archipelagos as ideal study systems where seabird tracking data offer a foundation for future research into how seamount features and their associated environmental conditions attract marine megafauna and facilitate the formation of biological hotspots. We advocate for global research and conservation initiatives to safeguard seamount biodiversity.

FIGURE 1. Information used to quantify seamount attractiveness for marine megafauna. Data sources can include environmental data (e.g., hydrodynamics and primary production), physical seamount features, fisheries data, and marine megafauna data.
> High res figure
|
PHYSICAL AND ECOLOGICAL DRIVERS OF SEAMOUNT BIODIVERSITY
Characterizing Seamounts: Physical Features, Oceanographic Processes, and Remote Sensing
Estimates of the global number of seamounts range from 10,000 to over 60,000 due to incomplete mapping and differences in algorithms that detect seamounts from bathymetry models (Yesson et al., 2021). Factors such as steepness, summit height and depth, geomorphology (e.g., conical, stellate, ridge-like, guyots, plateaus), and bottom depth are used to categorize seamounts, with digital terrain models from depth data employed to assess morphology (Cascão et al., 2017). Physical characteristics of seamounts influence biological processes by shaping nutrient distribution and organism aggregation. Seamounts with summit depths in the euphotic zone, where light levels support positive net photosynthesis, often host zooxanthellate corals and macroalgae communities that contribute to primary productivity (Yesson et al., 2011). Three-dimensional seamount structure, including slope steepness and overall morphology, further modifies productivity patterns by creating distinct hydrodynamic environments (White et al., 2007; Lavelle and Mohn, 2010; Rogers, 2018). Hydrodynamic processes interact with seamounts, forming internal waves, eddies, and localized upwelling, which enhance nutrient circulation and stimulate phytoplankton growth, thereby increasing productivity in surrounding waters (Boehlert and Genin, 1987; Lavelle and Mohn, 2010). Those seamounts with summit depths between the euphotic zone and approximately 1,500 m may associate with accumulation layers of zooplankton and micronekton (Genin and Dower, 2007). Additionally, topographic blocking occurs when seamounts prevent organisms from descending past the summit during diel vertical migration, regularly concentrating them at predictable depths (Isaacs and Schwartzlose, 1965; Genin, 2004). This productivity provides additional mid-level trophic enhancement of zooplankton and micronekton, attracting migratory pelagic predators, and emphasizing seamounts as biodiversity hotspots (Morato et al., 2008b). Hydrodynamic modeling can simulate complex flow patterns and physical processes at seamounts (Mohn et al., 2023). When coupled with agent-based models (Fulton et al., 2007), such approaches may help explain how prey fields make seamounts attractive to predators. Researchers use subsurface acoustic Doppler current profilers, active acoustics surveys, and visual techniques including autonomous and remotely operated underwater vehicles and baited cameras to study these processes and related productivity (Letessier et al., 2019; Ramiro-Sánchez et al., 2019).
Remote-sensing technologies have improved our ability to study seamount-biodiversity relationships by providing environmental data at relevant spatial and temporal scales (Leitner et al., 2020). Parameters, including sea surface temperature (SST), chlorophyll concentration, primary production, ocean currents, and shallow water bathymetry, are now estimated and analyzed with increased accuracy and greater global coverage (Mendonca et al., 2010; Oliveira et al., 2016; Leitner et al., 2020). Validated open-source data from global modeling and remote-sensing products, including those provided by the Copernicus Marine Environment Monitoring Service, allow researchers to construct comprehensive models of marine ecosystems, linking physical oceanographic features with biological productivity and animal behaviors. For instance, chlorophyll concentration serves as a proxy for phytoplankton abundance and primary production, indicating potential areas of high zooplankton and micronektonic prey availability for seabirds and identifying foraging hotspots (Santos et al., 2019). Ocean current and SST data help further our understanding of the formation of productive frontal systems and eddies (Cole and Villacastin, 2000), which concentrate prey at seamounts (Lavelle and Mohn, 2010; Mohn et al., 2021). Ongoing advancements in satellite technology, including the Behrenfeld method of estimating vertical migration (Behrenfeld et al., 2019), and new data processing techniques continue to improve assessments of seamount-driven ecosystem dynamics.
Fisheries’ Impacts on Seamount Ecosystems: Historical Patterns and Management Implications
Since the mid-twentieth century, seamounts have been targets for intensive fishing operations (Koslow et al., 2000) and harvesting of precious corals (such as Corallium rubrum; Simpson and Watling, 2011; Baco et al., 2023). Many species fished at seamounts have traits that render them slow to recover from overfishing, such as long lifespans, low fecundity, and limited dispersal (Koslow, 1997). These species often exist in low numbers but congregate at seamounts during specific life stages, like reproduction (Clark, 1996). The pattern of serially locating and depleting these aggregations raises significant concerns about the long-term viability of seamount fisheries (Koslow et al., 2000). There have been efforts to document the effects of seamount protection on fish abundance. Specifically, the Condor seamount observatory (Azores archipelago) was established to study seamount ecosystem dynamics and has been a crucial site for monitoring the effects of fishing cessation and conservation efforts (Giacomello et al., 2013). After nine years of fishing cessation, some species, like Pagellus bogaraveo, showed significant increases in abundance and biomass, indicating the effectiveness of protection measures (Giacomello et al., 2020). Fisheries-dependent data, including fishing locations, catch data, species composition, and abundance, offer insights into seamount attractiveness to target species and their predators and competitors (Morato et al., 2008b). Seabird bycatch rates near seamounts can indicate which areas are most frequented (Montrond, 2020). These analyses can reveal seasonal patterns and oceanographic drivers of productivity at seamounts, improving our understanding of complex food web dynamics around seamounts and why they serve as important foraging grounds. However, fisheries management regulations may complicate interpretation of patterns. For example, changes in catch limits can alter stock status and prey availability (Clark, 1996), while spatial closures can shift local fish abundance and distribution (Giacomello et al., 2020), potentially changing predator aggregation at seamounts through mechanisms unrelated to hydrodynamic and trophic processes that naturally concentrate prey.
Marine Megafauna Distribution Patterns Around Seamounts: Observational Studies and Tracking Research
Seamounts often act as hotspots for marine megafauna (Clark et al., 2010; Weber et al., 2025). Recent quantitative assessments demonstrate biomass enhancements across trophic levels at shallow seamount summits compared to pelagic baselines (Weber et al., 2025). Fishery observer program sighting data have been used to assess marine megafauna presence and abundance near Azorean seamounts. Several marine species were observed at increased rates near shallow seamount summits, suggesting that summit depth influences productivity and species aggregation (Morato et al., 2008b). Cetaceans, along with loggerhead sea turtles (Caretta caretta), have been observed in close proximity to seamount peaks, suggesting that these underwater features are important for multiple marine species (Fiori et al., 2016). Additionally, higher seabird density and biomass have been documented around seamounts compared to adjacent areas (Haney et al., 1995).
Advancements in tracking technology have revolutionized our ability to study links between animal movement and conservation needs (Nathan et al., 2022). Tracking seabird movements permits investigation of fine-scale habitat use around seamounts (Medrano et al., 2023). These technologies have revealed that many seabird species reliably appear in specific areas and time periods at the macroscale (Frederiksen et al., 2016), often congregating in habitats or migratory corridors associated with oceanographic features that enhance productivity (Morten et al., 2025). Global Positioning System (GPS) tracking technology allows researchers to distinguish foraging using ground speed and movement directionality and to analyze detailed environmental factors that make seamounts attractive for marine megafauna. Recent work reveals that distance from seamounts predicts foraging behavior and habitat use in multiple procellariiform species (petrels, shearwaters, albatrosses, and diving petrels; Neves et al., 2023; Ventura et al., 2024).
We focus on the Cabo Verde and Azores archipelagos, where GPS tracking of seabirds provides fine-scale data on habitat use in areas surrounding specific seamounts. More specifically, tracking data in Cabo Verde reveal that several seabird species concentrate activity over seamount areas (authors González-Solís and Paiva, unpublished data; reviewed in Orejas et al., 2025). In the Azores, seamount-related features emerge as strong predictors of where seabirds forage (Neves et al., 2023). These two case studies illustrate how seabird tracking data can be used to link foraging behavior to specific seamount-related factors and to help identify areas of conservation importance.
CASE STUDY 1. Cabo Verde
The Cabo Verde archipelago, located in the North Atlantic Ocean off West Africa, has at least 14 major seamounts (including Senghor, Boa Vista, Cabo Verde, Maio, Cadamosto, and Nola; Orejas et al., 2025) within its exclusive economic zone (EEZ). Islands and seamounts form a horseshoe pattern opening westward in two chains. Eastern seamounts show age and erosion, while western seamounts display younger morphologies (Kwasnitschka et al., 2024). Interactions between the Canary Current, North Equatorial Current, North Equatorial Counter-Current, and Mauritanian Current foster a diverse ecosystem (Mittelstaedt, 1991). Complex bathymetry and mesoscale eddies create internal waves that enhance productivity above seamount summits (Mohn et al., 2021) and provide abundant food for benthic communities (Vinha et al., 2024). Reef-building species contribute to biodiversity and may harbor undiscovered species (Ramiro-Sánchez et al., 2019; Weber et al., 2025).
Cabo Verde waters contain hundreds of fish species that exhibit a high degree of endemism, making the area a priority for marine conservation (Wirtz et al., 2013). Mesopelagic and nektonic communities around Senghor Seamount undergo diel vertical migrations, becoming accessible to surface-feeding predators during their nighttime ascents from deeper waters (Mohn et al., 2021). Seamount-generated upwellings support complex food webs and serve as aggregation sites for pelagic species like tuna (Thunnus spp.) and billfish (Morato et al., 2010). These areas provide habitats for endemic and commercially valuable fish species (Wirtz et al., 2013), a pattern observed at many seamounts (Shank, 2010). The Cabo Verde region hosts large shark and ray populations, including IUCN red-listed species (species at risk of extinction; Montrond et al., 2020), and supports multiple cetacean populations (Hazevoet et al., 2010). The archipelago lies along migratory routes for humpback whales (Megaptera novaeangliae; Hazevoet et al., 2010) and sea turtles (Marco et al., 2011) and is an important breeding site for loggerhead sea turtles (Caretta caretta; Roast et al., 2023).
Long-term monitoring efforts have provided robust datasets on several breeding seabird species (Figure 2), including five endemic taxa: Cape Verde shearwater (Calonectris edwardsii), Cape Verde petrel (Pterodroma feae), Cape Verde storm-petrel (Hydrobates jabejabe), Cape Verde little shearwater (Puffinus boydi), and white-faced storm-petrel subspecies Pelagodroma marina eadesorum (Semedo et al., 2021; Medrano et al., 2023). These populations face threats from fishing bycatch, habitat degradation, invasive predators, light pollution, and human disturbance (Semedo et al., 2021). GPS tracking reveals that Cape Verde shearwaters make long foraging trips to productive waters off Dakar, Senegal (Paiva et al., 2015). The Cape Verde petrel undertakes trans-equatorial migrations between the Azores and Brazilian coastal waters during the non-breeding period (Ramos et al., 2017).
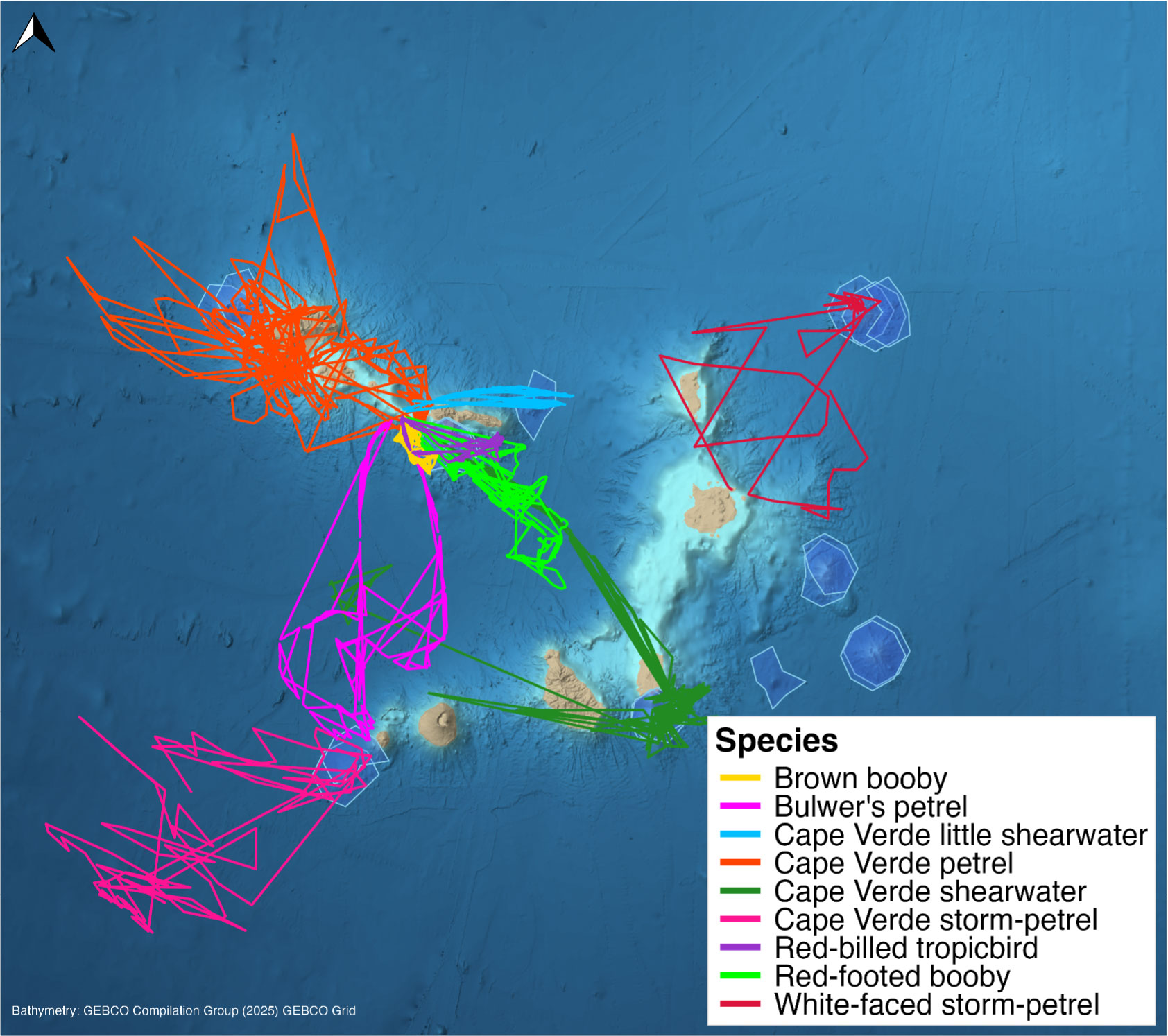
FIGURE 2. Examples of tracks from multiple seabird species observed around seamounts in the Cabo Verde archipelago. Scientific names of seabird species from top to bottom in the map legend: Sula leucogaster, Bulweria bulwerii, Puffinus boydi, Pterodroma feae, Calonectris edwardsii, Hydrobates jabejabe, Phaethon aethereus, Sula sula, and Pelagodroma marina. Seamounts are indigo polygons (Yesson et al., 2021). Tracks shown represent the nine species for which GPS data were available. Map created in R 4.5.1 (R Core Team, 2025).
> High res figure
|
GPS tracking datasets on Cabo Verde seabirds have grown in recent years (Paiva et al., 2015; Militão et al., 2017; dos Santos et al., 2023; Orejas et al., 2025), providing the spatio-temporal resolution to begin identifying which seamount features and surrounding conditions attract foraging seabirds. When tracks from multiple species are mapped together, GPS fixes frequently cluster over seamount projections (Figure 2; González-Solís and Paiva, unpublished data, referenced in Orejas et al., 2025). Cape Verde petrels, white-faced storm-petrels, and Bulwer’s petrels usually forage after sunset and at night (Dias et al., 2016; Ramos et al., 2017; Medrano et al., 2023) and likely do not associate with fishing vessels (Montrond, 2020; Almeida, 2021; Navarro-Herrero et al., 2025), suggesting their concentration over seamount projections reflects a response to natural prey availability (Orejas et al., 2025). These species may target micronekton aggregations driven toward the surface by DVMs (Carreiro et al., 2023; dos Santos et al., 2023), a process observed at Senghor Seamount (Mohn et al., 2021; Orejas et al., 2025). Cape Verde shearwaters have been recorded visiting the projections of Maio Seamount and João Valente slopes. However, they are diurnal foragers that associate with fishing vessels (Montrond, 2020; Almeida, 2021), and tuna at seamounts may also drive prey to the surface, creating additional foraging opportunities (Orejas et al., 2025). These overlapping factors make it difficult to isolate the role of seamount processes in attracting this species. Diet and tracking analyses reveal distinct foraging strategies between sympatric Cabo Verde seabirds (dos Santos et al., 2023), indicating that these features may provide a range of resources used by several species. These tracking datasets, combined with seamount-related environmental data, offer a path toward understanding why some areas around seamounts are more attractive to foraging seabirds than others.
Cabo Verde has established 17 marine/coastal protected areas (Decree-Law No. 3/2003, UNDP, 2009; Decree-Law No. 44/2006, National Directorate for the Environment, 2015, reviewed in Larrea et al., 2023). The archipelago hosts a rich assemblage of marine species, and seabird tracking data are now providing the spatial detail needed to protect them. Tracking data have contributed to the designation of marine Key Biodiversity Areas and informed a protection proposal for waters surrounding the islands of Fogo and Brava and the Rombos islets, including the Cadamosto Seamounts (Orejas et al., 2025). As tracking coverage expands, these data have the potential to guide how the protection of seamount areas is designed and prioritized.
CASE STUDY 2. The Azores
The Azores archipelago, in the central North Atlantic, contains 68 large and 398 small seamount-like features that occupy 37% of its EEZ (Morato et al., 2008a). Prominent seamounts include Condor, Gigante, Dom João de Castro, Princess Anna, and Princess Alice. Though many seamounts occur in chains, isolated seamounts are also present. Considerable variations in size and shape prevent generalizations regarding morphology (Morato et al., 2008a).
The region’s complex oceanography, influenced by the Azores Current system, creates a dynamic environment that supports diverse marine life (Mohn et al., 2023), and its seamounts serve as biodiversity hotspots with enhanced productivity. Diel vertical migrations that concentrate micronekton near the surface over seamount summits increase prey biomass at night (Cascão et al., 2017, 2019). Benthic and benthopelagic fish rely on this prey, highlighting trophic connections between mesopelagic and deeper communities (Colaço et al., 2013). This increased micronektonic prey biomass attracts predators such as tuna, seabirds, and marine mammals to the area (Morato et al., 2008b). Seamounts support diverse deep-sea coral and sponge communities (Morato et al., 2013), and numerous pelagic and demersal fish species are closely associated with seamount habitats (Afonso et al., 2020). The area hosts 24 whale and dolphin species (Silva et al., 2014) and is an important foraging and breeding site for seabirds (Figure 3), including Cory’s shearwater (Calonectris borealis) and the endemic Monteiro’s storm-petrel (Hydrobates monteiroi).
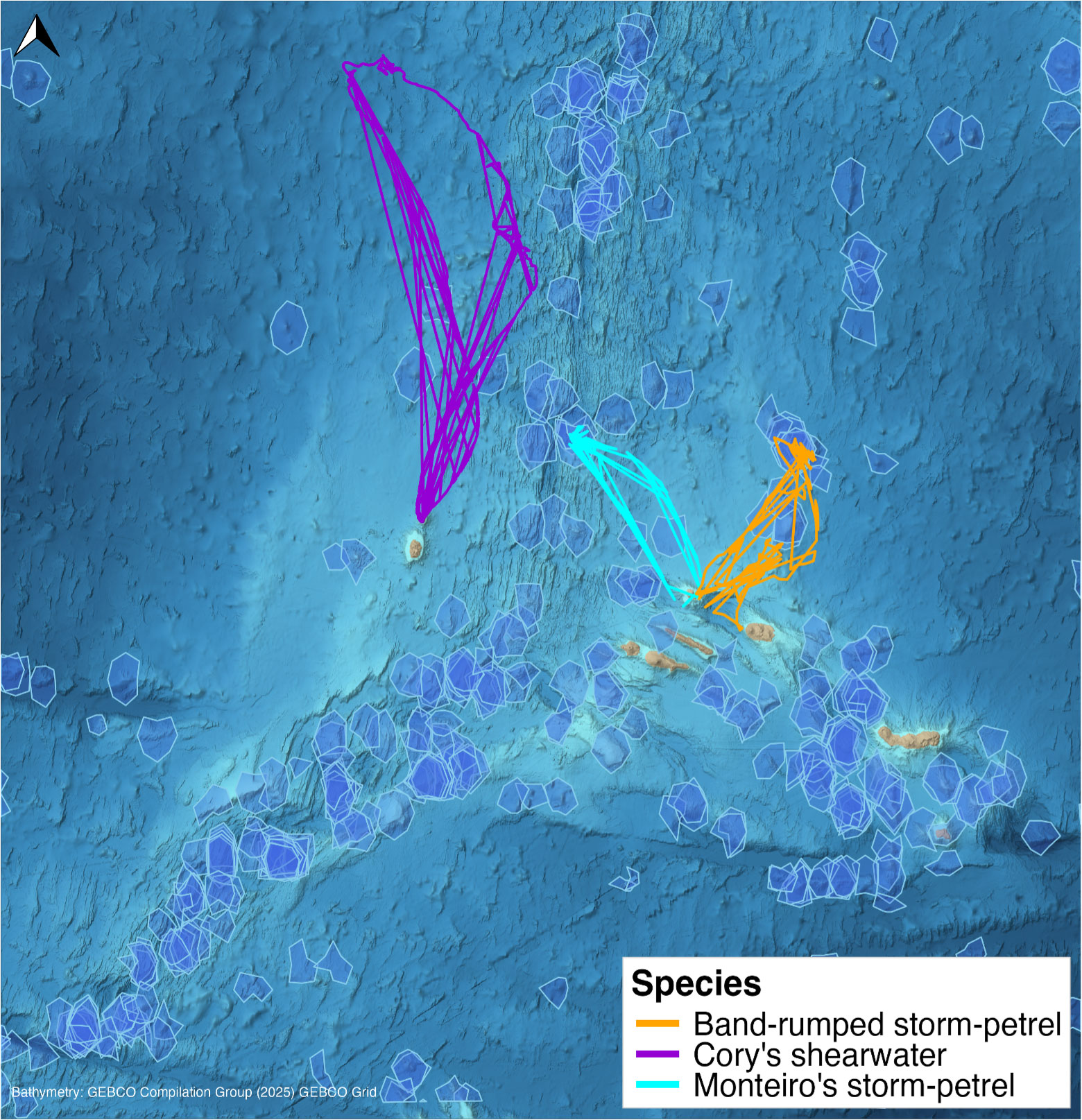
FIGURE 3. Examples of tracks from multiple seabird species observed around seamounts in the Azores archipelago. Scientific names of seabird species from top to bottom in the map legend: Hydrobates castro, Calonectris borealis, and Hydrobates monteiroi. Seamounts are indigo polygons (Yesson et al., 2021). Tracks shown represent three species for which GPS data were accessible. Map created in R 4.5.1 (R Core Team, 2025).
> High res figure
|
Fine-scale GPS tracking data from seabird colonies in the Azores offer an opportunity for future work to explore the relationship between seamount-related characteristics and seabird foraging. Cory’s shearwaters from Corvo Island were found to commute to a seamount-rich region of the Mid-Atlantic Ridge, where depth gradients and productivity fronts influenced residence time at foraging sites (Paiva et al., 2010). Neighboring sub-colonies on Corvo also partitioned foraging space at sea, demonstrating that habitat use varied across small spatial scales (Ceia et al., 2015). Tracking data from Monteiro’s storm-petrel on Graciosa Island further link seabird foraging to seamount features. Foraging locations were most strongly predicted by seafloor depth and proximity to the nearest seamount, with sea surface temperature and chlorophyll concentration as secondary factors, and foraging areas remained consistent across four breeding seasons (Neves et al., 2023). These findings show that tracking data can be used to help identify which seamount areas attract foraging seabirds, though the range of environmental variables tested so far has been limited. GPS coverage currently spans a few species (Figure 3), and expanding to additional taxa would reveal whether these foraging grounds are shared more broadly. Such data carry conservation implications. For example, most tracked Monteiro’s storm-petrel foraging locations were found to be unprotected, with only 11.4% falling within designated MPAs, despite 95% of unprotected locations being located within the Azores EEZ where national management is possible (Neves et al., 2023).
In 2024, the Azores approved legislation to expand the Azores Marine Park and establish the largest MPA network in the North Atlantic; to preserve the archipelago’s biodiversity and support sustainable use of marine resources, fishing and other damaging activities are limited there (Regional Legislative Decree No. 28/2011/A, updated version; also Tojeira et al., 2025). Multi-species predictive models that combine data on tracking, seamount features, and environmental variables can help evaluate whether this network adequately covers areas of high ecological value (Neves et al., 2023). Thus, the Azores offer an opportunity to test whether formal protection changes how marine predators use seamount areas over time.
DISCUSSION AND FUTURE DIRECTIONS
Understanding what makes seamounts attractive to marine megafauna can serve as a useful conservation tool. Cabo Verde and the Azores provide excellent locations for research that investigates factors influencing megafauna aggregation. Hydrodynamic conditions can create heterogeneous habitats that attract megafauna (Boehlert, 1988; Godø et al., 2012; Annasawmy et al., 2019), though not all seamount areas function as biodiversity hotspots. For example, seamounts with shallow to intermediate depths, steep slopes, and proximity to other seamounts show chlorophyll enhancements that support trophic enhancement in some areas (Leitner et al., 2020). Conversely, there is limited biological enhancement at isolated seamounts with deep summits and minimal current interactions. Understanding seamount ecological value to prioritize conservation efforts requires multi-species predictive models that identify biological hotspots and indicate how hydrodynamic features support different life stages of marine species. Here, we pose six questions regarding key knowledge gaps that, once addressed, will advance this understanding and support the conservation of important seamount ecosystems.
- At which spatial and temporal scales do seamounts influence physical and ecological processes?
Seamount observational studies are often limited in scale, making it unclear how they shape biogeography from local to regional spatial scales and daily to multi-year timescales. Current knowledge relies largely on episodic ship surveys and surface satellite data, which do not capture subsurface trophic dynamics or temporal variability. Sustained, depth-resolved observing systems at representative seamounts, alongside predictive models integrating physical oceanography with biological responses across nested scales, are needed to enhance understanding of seamount roles in marine ecosystems.
- Why are some seamounts biodiversity hotspots while others are not?
We lack a comprehensive understanding of how seamount location, topography, and oceanographic conditions interact to enhance productivity and biodiversity. Integrated analyses of hydrodynamics, nutrient fluxes, primary productivity, and species assemblages are needed to determine why some seamounts attract life across trophic levels while others remain relatively barren.
- What role do seamounts play in enhancing ecological connectivity in the open ocean?
While seamounts act as steppingstones for species dispersal, gene flow, and migration, their role in connecting isolated habitats remains poorly understood. Studying mechanisms that facilitate connectivity will elucidate their importance for population resilience and biodiversity networks.
- How do functional roles of seamounts vary across marine megafauna taxa?
Many large marine species, including sea turtles, marine mammals, seabirds, and elasmobranchs, associate with seamounts as foraging hotspots, migratory waypoints, or breeding grounds, yet drivers of these behaviors remain unclear. Seabirds, for which fine-scale tracking data are particularly abundant, offer a practical starting point for linking specific seamount features to behavior. Understanding which seamount features (e.g., topography, productivity, prey availability, isolation, seabed habitat complexity) promote specific behaviors across taxa and life stages is important for targeted conservation.
- How resilient are seamount ecosystems to environmental change and human impacts?
These ecosystems face pressures from climate change, deep-sea mining, overfishing, and pollution, yet their capacity to endure and recover remains largely unknown. Studies on species turnover, habitat degradation, and adaptive responses are needed to predict ecological shifts and support adaptive management.
- Which seamounts should be prioritized for protection?
A science-based framework is needed to identify seamounts of high conservation value based on biodiversity richness, ecological connectivity, functional resilience, and vulnerability to human activities. Such criteria can guide MPA design and management strategies.
A mechanistic understanding of interactions between marine megafauna and seamounts, particularly considering complex hydrodynamic processes, will provide stakeholders with tools to identify and prioritize seamounts for protection. Specifically, the Cabo Verde and Azores case studies show how seabird tracking data can reveal these relationships at a fine scale. Such insights can support the development of area-based management strategies like MPAs and marine spatial planning. At the international level, the new BBNJ Agreement defines the processes and instruments for establishing MPAs in areas beyond national jurisdiction where most seamounts occur. The Agreement proposes criteria to help identify candidate MPAs based on ecological significance. Investigating how megafauna interact with seamounts aligns such criteria by assigning special importance for species using seamounts; benefiting threatened, endangered, or declining species; and facilitating important ecological processes. At the national level, marine spatial planning processes, like those in the Azores, similarly require identifying hotspots of seamount-megafaunal interactions and their underlying mechanisms. This information contributes to evaluating whether existing management measures ensure conservation and sustainable use and identifies related gaps in policy and technical measures (Afonso et al., 2020). The recent proposal to establish the Reserva Natural Marinha D. Carlos, encompassing approximately 173,000 km² of Northeast Atlantic seamount complexes in Portuguese waters (Order No. 12518/2025; Portuguese Government, 2026), illustrates growing policy interest in seamount protection. Our proposed framework for research that identifies attractive seamounts supports conservation objectives at both national and international scales by providing objective prioritization criteria. Research on seamount ecosystem complexities will enable informed conservation measures to safeguard these major components of ocean biodiversity for future generations.
ACKNOWLEDGMENTS
L. Broadus and C. Mohn acknowledge funding by a VILLUM FONDEN (Denmark) research grant (VIL58674, “A global assessment of seabird and seamount connections”). F. Ventura acknowledges support by the National Aeronautics and Space Administration (NASA), grant 80NSSC25K7836. This work received national funds through the FCT – Foundation for Science and Technology, I.P., under the project UIDB/05634/2025 and UIDP/05634/2025 and through the Regional Government of the Azores through the project M1.1.A/FUNC.UI&D/003/2021-2024.





